Acid Rain and the Environment
Effects on the environment:
Acid Rain can effect different parts of the environment:
- Forests: Acid rain can damage parts of forests. It may destroy trees and plants, including significant primary producers, therefore, acid rain can affect entire ecosystems. Primary producers are significant in an ecosystem, because they provide food for animals. Therefore, acid rain does not only affect single trees and plants, but entire ecosystems. Ecosystems are systems that are made up of organisms and their interactions among each other. Acid rain can decrease the speed at which trees and plants grow. Furthermore, acid rain dissolves and washes the calcium in the soil and in the leaves away, which is necessary for a tree to grow. Acid rain additionally washes away other nutrients and vitamins, that are vital for a tree to grow. Therefore, the trees grow at a slower rate. The lack of nutrients and vitamins also causes the tree to become more vulnerable and sensitive to the cold, insects and diseases. Acid rain releases aluminum into the soil, which can end up in aquatic environments, such as streams rivers and lakes, too, where it can harm the organisms.
-Aquatic Life: Acid rain also has the largest impact on streams, lakes and marshes. Most lakes have a pH ranging between 6 and 8. Soil is able to neutralize slightly acidic rainwater, which is not caused by pollution, but if the rainwater is so acidic, the soil is not able to and the lake becomes acidic. Although some animals can survive in acidic water, many animals are either forced to leave their habitats or they die from the acidic conditions in their surrounding. Fish cannot swim in acidic lakes, nor hatch eggs. In addition, aluminum can wash into the lakes, streams or marshes, making the water toxic for many aquatic animals.
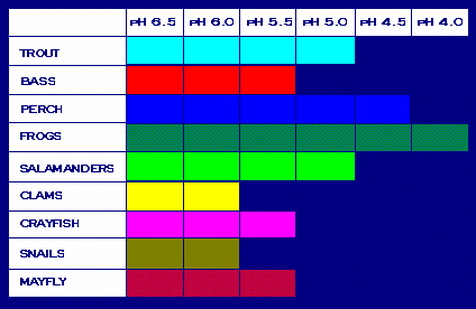
The graph on the right, which was created by the Environmental Protection Agency (EPA), shows what animals can survive until which pH levels. One can see that several aquatic animals cannot survive if the pH gets lower, so more acidic. Therefore, many animals will not survive in acidic lakes. This can harm the entire ecosystem since animals are reliant on each other since they feed each other. If one species dies out or leaves, another species might have less food supply.
The graph on the right, which was created by the Environmental Protection Agency (EPA), shows what animals can survive until which pH levels. One can see that several aquatic animals cannot survive if the pH gets lower, so more acidic. Therefore, many animals will not survive in acidic lakes. This can harm the entire ecosystem since animals are reliant on each other since they feed each other. If one species dies out or leaves, another species might have less food supply.
What is being done:
Most importantly, in order to reduce acid rain, the emission of gases has to be limited and minimized. Since sulfur dioxide and nitrogen oxides are released during the process of fuel combustion and in factories in general. In the past, the governments of different countries have tried to reduce the emissions of the gases that cause acid rain, sulfur dioxide and nitrogen oxides. Governments are requiring that all power plants clean their smoke stacks, which means that some of the harmful gases in smoke stacks are trapped so that they do not enter the atmosphere and cause acid rain. Governments have also tried promoting alternative and renewable energy sources, that do not burn fossil fuels whilst creating energy. There are many different types of alternative energy sources, which do not emit sulfur dioxide or nitrogen oxides during the production of energy, for example, solar power, wind energy or hydropower.
In the USA, the Environmental Protection Agency (EPA) is responsible for reducing the amount of pollutant gases emitted. In 1963 the Clean Air Act was passed, which included limits for harmful pollutants. In 1977 many states did not meet the standards of the Clean Air Act. Therefore, a new amendment was added to the Clean Air Act stating that older industrial factories would have to be assessed in order to be used. In 1990, more amendments were passed, which included the Acid Rain Program, which is lead by the EPA. According to the EPA, "the overall goal of the program is to achieve significant environmental and health benefits through reductions in emissions of sulfur dioxide (SO2) and nitrogen oxides (NOx), the primary causes of acid rain." (EPA) Basically, the Acid Rain Program sets up limits of how much SO2 and NOx a power plant or factory can emit annually. In order to minimize the emission of SO2 gases, the Acid Rain Project has created a "cap and trade program", which is a regulatory system, Additionally, the NOx is reduced using a rate based regulatory system. Since 1990, the emissions of sulfur dioxide were reduced 33% and the emissions of nitrogen oxides by approximately 27%. (EPA) These statistics show that the Acid Rain Program has clearly contributed to the reduction of pollutants and minimized the amount of gases emitted in many companies. Furthermore, the amount of acid rain in the USA has decreased by 30% since the Acid Rain Program has started.
The USA is not the only place in which acid rain has clearly had affects on the environment. Europe is also affected by this major environmental issue, acid rain. Also Europe tried to minimize the emissions of SO2 and NOx with different protocols signed by different countries. In 1985 the majority of the members of the United Nations Economic Commission for Europe agreed to reduce the emissions of sulfur dioxide from the year 1980 by 30% until 1993. All countries that agreed to sign the protocol managed to reduce their SO2 emissions by at least 30%. In 1994 several European countries agreed to reduce their SO2 emissions from 1980 by 70-80% by the year 2000. The east European countries set teir goal to 40-50%. Almost all countries managed to reduce their SO2 emissions by the target amount. From 1993 onward, all cars included a catalytic converter, which also minimized the pollution emitted by a car and the gases emitted. Generally, the SO2 emissions were already reduced greatly, while the NOx emissions reduce at a slower rate.
As aforementioned, scientists have developed several techniques to reduce the emission of gases during coal combustion. These methods include: low sulfur coal, scrubbers and fluidized bed combustion.
In the USA, the Environmental Protection Agency (EPA) is responsible for reducing the amount of pollutant gases emitted. In 1963 the Clean Air Act was passed, which included limits for harmful pollutants. In 1977 many states did not meet the standards of the Clean Air Act. Therefore, a new amendment was added to the Clean Air Act stating that older industrial factories would have to be assessed in order to be used. In 1990, more amendments were passed, which included the Acid Rain Program, which is lead by the EPA. According to the EPA, "the overall goal of the program is to achieve significant environmental and health benefits through reductions in emissions of sulfur dioxide (SO2) and nitrogen oxides (NOx), the primary causes of acid rain." (EPA) Basically, the Acid Rain Program sets up limits of how much SO2 and NOx a power plant or factory can emit annually. In order to minimize the emission of SO2 gases, the Acid Rain Project has created a "cap and trade program", which is a regulatory system, Additionally, the NOx is reduced using a rate based regulatory system. Since 1990, the emissions of sulfur dioxide were reduced 33% and the emissions of nitrogen oxides by approximately 27%. (EPA) These statistics show that the Acid Rain Program has clearly contributed to the reduction of pollutants and minimized the amount of gases emitted in many companies. Furthermore, the amount of acid rain in the USA has decreased by 30% since the Acid Rain Program has started.
The USA is not the only place in which acid rain has clearly had affects on the environment. Europe is also affected by this major environmental issue, acid rain. Also Europe tried to minimize the emissions of SO2 and NOx with different protocols signed by different countries. In 1985 the majority of the members of the United Nations Economic Commission for Europe agreed to reduce the emissions of sulfur dioxide from the year 1980 by 30% until 1993. All countries that agreed to sign the protocol managed to reduce their SO2 emissions by at least 30%. In 1994 several European countries agreed to reduce their SO2 emissions from 1980 by 70-80% by the year 2000. The east European countries set teir goal to 40-50%. Almost all countries managed to reduce their SO2 emissions by the target amount. From 1993 onward, all cars included a catalytic converter, which also minimized the pollution emitted by a car and the gases emitted. Generally, the SO2 emissions were already reduced greatly, while the NOx emissions reduce at a slower rate.
As aforementioned, scientists have developed several techniques to reduce the emission of gases during coal combustion. These methods include: low sulfur coal, scrubbers and fluidized bed combustion.
What YOU can do as an individual:
Not only the governments and scientists can solve this issue; however, you are also capable of contributing to the reduction of acid rain. There are several things that you can do in order to help reduce acid rain and help preserve our environment. Most importantly, conserving energy is an efficient way of reducing acid rain since the less energy is used, the less energy has to be produced. Therefore, fewer power plants have to be used, power plants have to create less energy and less fossil fuels have to be burned. Therefore, the emission of sulfuric and nitric acid is decreased. Another way is finding an alternative to a car in order to decrease the amount of pollution made. In order to do this, use other modes of transport, such as public transport (train, bus, underground, etc.), carpooling, biking or even walking short distances instead of using your car.

