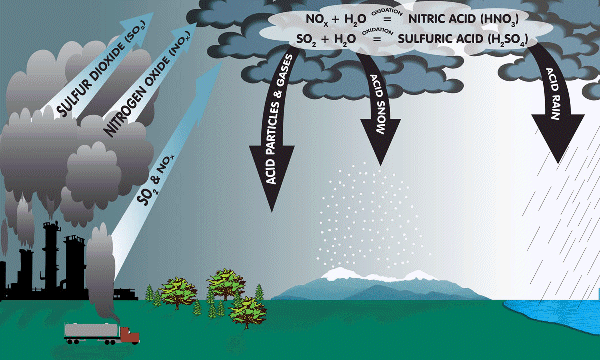
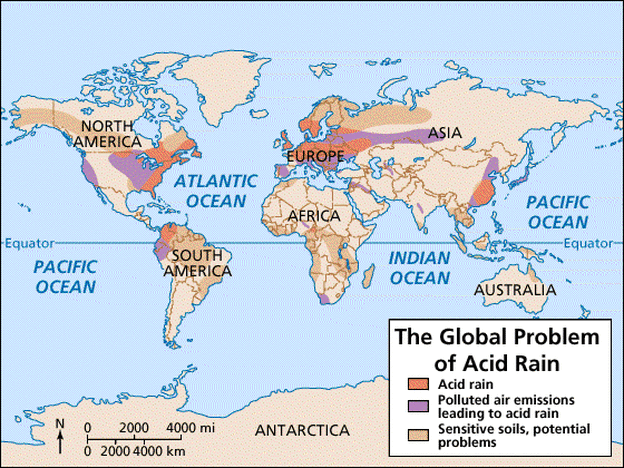
What is Acid Rain?
Acid rain is any kind of precipitation containing an abnormal high amount of sulfuric or nitric acid. Acid rain results in chemicals being released into the atmosphere, either due to natural sources, but most of all human activity. Volcanoes and decaying vegetation cause acids to be released into the air, but humans cause the highest emissions of sulfur dioxide and nitrogen oxides. Acid rain is primarily caused by the emission of pollutant gasses, which are released by electric power stations or other power plants, that are responsible for fossil fuel combustion. Acid rain is a widespread issue, which is causing damage to soil, water chemistry, and the life cycles of plants and animals on land and in the water.
How is Acid Rain formed?
During the process of fossil fuel combustion, which is the burning of coal, oil or other natural gas, the chemical gases sulfur dioxide (SO2) and nitrogen oxides (NOx) are emitted into the air. When these gases react with water, oxygen and other chemicals, the reaction results in a mild solution of sulfuric and nitric acid. Sulfur dioxide becomes sulfuric acid, while the nitrogen oxides become nitric acid (SO2 --> H2SO4 and NOx --> HNO3). The acids can be moved throughout the atmosphere easily for over hundreds of miles due to the wind. Therefore, acid rain can also fall away from the source of pollution. While the sulfuric and nitric acids travel, they become a part of rain sleet, snow and fog. After this process, the precipitation is more acidic than normally. Another term for acid rain is acidic deposition. There are two types of acidic deposition, wet and dry. Wet deposition is considered any type of precipitation that reduces the acids in the atmosphere by depositing them on the earths surface. Dry deposition occurs when polluting particles or gases combined with dust or smoke become acidic.
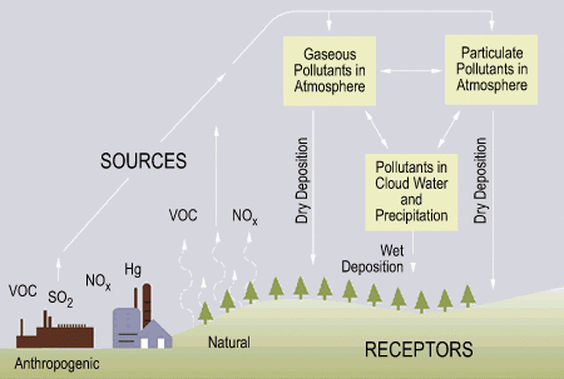
- The pollutant gases (So2, NOx) are emitted from the sources, i.e. a power plant burning fossil fuels.
- Gases are released into the atmosphere.
- The gases combine with dust or smoke and become acidic or particulate pollutants are formed and dry deposition occurs.
- Gases react with water and oxygen and therefore, become acidic.
- Wet deposition onto the earth's surface occurs through a type of precipitation, i.e. rain, snow. etc.
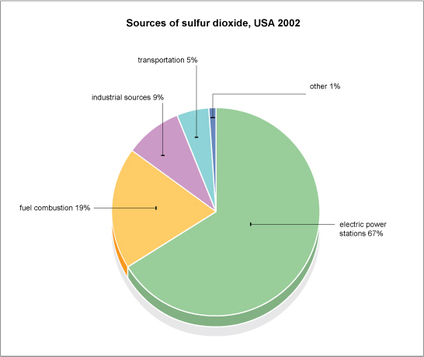
The pie chart on the right, which was created by the BBC, shows what sources emit the most amounts of sulfur dioxide. According to the chart, electric power stations release the most SO2 into the atmosphere, followed by fuel combustion. Industrial sources and transportation emit a smaller amount of SO2 into the atmosphere.